UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
FORM 8-K
________________
CURRENT REPORT
PURSUANT TO SECTION 13 OR 15(d)
OF THE SECURITIES EXCHANGE ACT OF 1934
Date of Report (Date of Earliest Event Reported) – January 10, 2007
________________________
WEST PHARMACEUTICAL SERVICES, INC.
(Exact name of registrant as specified in its charter)
_____________________
|
|
|
|
|
Pennsylvania |
1-8036 |
23-1210010 |
|
(State or other jurisdiction |
(Commission File Number) |
(IRS Employer Identification No.) |
|
|
|
|
|
101 Gordon Drive, PO Box 645, Lionville, PA |
|
19341-0645 |
|
(Address of principal executive offices) |
|
(Zip Code) |
610-594-3319
(Registrant’s telephone number, including area code)
Not Applicable
(Former name or address, if changed since last report)
____________________
Check the appropriate box below if the Form 8-K filing is intended to simultaneously satisfy the filing obligation of the registrant under any of the following provisions:
|
o |
Written communications pursuant to Rule 425 under the Securities Act (17 CFR 230.425) |
|
o |
Soliciting material pursuant to Rule 14a-12 under the Exchange Act (17 CFR 240.14a-12) |
|
o |
Pre-commencement communications pursuant to Rule 14d-2(b) under the Exchange Act (17 CFR 240.14d-2(b)) |
|
o |
Pre-commencement communications pursuant to Rule 13e-4(c) under the Exchange Act (17 CFR 240.13e-4(c)) |
Kthelman/SEC drafts/Form 8-K - January 2007 CJS Conference
Item 7.01 Regulation FD Disclosure
On January 10, 2007, West Pharmaceutical Services, Inc. (the “Company”) issued a press release updating its expected 2006 sales and its expected earnings per share for the fourth quarter and year ended December 31, 2006. The release also noted that Donald E. Morel, Jr., Ph.D., the Company’s Chairman and Chief Executive Officer, and William J. Federici, the Company’s Chief Financial Officer, will be presenting at the CJS Securities Investor Conference on January 11, 2007. A copy of the press release is attached hereto as Exhibit 99.1 and incorporated herein by reference.
Attached hereto as Exhibit 99.2 and incorporated herein by reference, is the Corporate Overview (Investor Presentation) to be given by Dr. Morel and Mr. Federici at the conference.
The information in this report (including Exhibit 99.1 and 99.2) is being furnished pursuant to Item 7.01 Regulation FD and shall not be deemed to be “filed” for purposes of Section 18 of the Securities Exchange Act of 1934 (the “Exchange Act”) or otherwise subject to the liabilities of that section, nor shall it be deemed to be incorporated by reference in any filing under the Securities Act of 1933 or the Exchange Act.
Item 9.01 Financials Statement and Exhibits
| (d) |
Exhibits |
|
|
|
|
|
|
|
|
|
Exhibit # |
|
Description |
|
|
99.1 |
|
West Pharmaceutical Services, Inc. Press Release, dated January 10, 2007. |
|
|
|
|
|
|
|
99.2 |
|
West Pharmaceutical Services, Inc. Corporate Overview (Investor Presentation). |
SIGNATURE
Pursuant to the requirements of the Securities Exchange Act of 1934, the registrant has duly caused this report to be signed on its behalf by the undersigned hereunto duly authorized.
| WEST PHARMACEUTICAL SERVICES, INC. |
|
|
| /s/ John R. Gailey III |
| John R. Gailey III |
| Vice President, General Counsel and Secretary |
January 10, 2007

|
Contacts: West Pharmaceutical Services, Inc. Michael A. Anderson Vice President and Treasurer (610) 594-3345 |
Investors/Financial Media: Financial Dynamics Julie Huang/Theresa Kelleher (212) 850-5600 wst@fd-us.com |
Exhibit 99.1
West Pharmaceutical Services, Inc. Updates Fourth Quarter and Full-Year 2006 Expectations
-Expects 2006 Earnings per Diluted Share from Continuing Operations between $1.90 and $1.94 -
-Company Sets Earnings Release Date –
-Company to Present at the CJS Investor Securities Conference -
LIONVILLE, Pa., January 10, 2007 – West Pharmaceutical Services, Inc. (NYSE: WST), the global market leader in closure systems and syringe components for use with injectable drugs, today updated its expected 2006 sales and expected earnings per share for the fourth quarter and year ended December 31, 2006. Consistent with its normal practice, the Company will report final fourth quarter and full year 2006 financial results on Tuesday, February 20, 2007, at which time management will discuss the business and financial outlook for 2007.
“Fourth quarter sales were slightly ahead of our earlier outlook and should yield earnings per share consistent with or just ahead of those expectations” said Donald E. Morel, Jr., Ph.D., West’s Chairman and Chief Executive Officer. “While we are awaiting the completion of the analyses of last year’s record results, management is very much focused on executing our operating and expansion plans for 2007, which we expect will be another year of strong sales and earnings growth.”
The Company now expects to report sales for the year 2006 of approximately $913 million. Excluding the effects of the items described below, earnings per diluted share from continuing operations are expected to be between $0.39 and $0.43 for the fourth quarter, and between $1.90 and $1.94 for the full year ended December 31, 2006. The expected results are based upon preliminary information and remain subject to revision when the Company concludes its normal period-end procedures and analyses. The expected annual results exclude an $0.11 per share first quarter 2006 charge for the cost of the early retirement of debt, net of an unrelated tax benefit. The expected annual results include the $0.03 net adverse impact, reported in the third quarter, of the costs associated with an impairment charge, a change in tax reserves, and an affiliated company’s asset-related charge.
West to Present at 6th Annual CJS Investor Conference
Donald E. Morel, Jr. Ph.D., the Company’s Chairman and Chief Executive Officer, and William J. Federici, Chief Financial Officer will present at an investor conference being sponsored by CJS Securities on Thursday, January 11, 2007. A copy of the presentation can be found in the “Investor” section of the Company’s website, www.westpharma.com.
West to Host Earnings Conference Call
The Company will report final fourth quarter and fiscal 2006 results on Tuesday, February 20, 2007 at 9:00 a.m. Eastern Time. A live broadcast of the conference call will be available at the Company’s web site, http://www.westpharma.com/, in the “Investor” section. Please allow extra time prior to the call to visit the site and download the streaming media software required to listen to the Internet broadcast.
About West Pharmaceutical Services, Inc.
West Pharmaceutical Services, Inc. is the world's premier manufacturer of components and systems for injectable drug delivery, including stoppers and seals for vials, and closures and disposable components used in syringe, IV and blood collection systems. The Company also provides products with application to the personal care, food and beverage markets. West's customers include the world's leading pharmaceutical, biotechnology, generic drug and medical device producers. Headquartered in Lionville, Pennsylvania, West Pharmaceutical Services supports its partners and customers from 50 locations throughout North America, South America, Europe, Mexico, Japan, Asia and Australia. For more information, visit West at http://www.westpharma.com.
Safe Harbor Statement
This press release contains some forward-looking statements that set forth anticipated results based on management’s plans and assumptions. Such statements give our current expectations or forecasts of future events – they do not relate strictly to historical or current facts. In particular, these include statements concerning preliminary financial results for which we have not yet completed all of the necessary analyses and reports in accordance with generally accepted accounting principals (GAAP). We have tried, wherever possible, to identify such statements by using words such as “estimate,” “expect,” “intend,” “believe,” “plan,” “anticipate” and other words and terms of similar meaning in connection with any discussion of future operating or financial performance or condition.
We cannot guarantee that any forward-looking statement will be realized. If known or unknown risks or uncertainties materialize, or if underlying assumptions are inaccurate, actual results could differ materially from past results and those expressed or implied in any forward-looking statement. You should bear this in mind as you consider forward-
looking statements. We cannot predict or identify all such risks and uncertainties, but factors that could cause the actual results to differ materially from expected and historical results include the following: Events occurring between the close of the year 2006 and the publication date of our financial statements for periods ending in 2006 that have a material effect and which are required to be given effect or disclosed in those financial statements; corrections of errors or changes to estimates based upon facts and circumstances discovered in the course of our periodic analyses and financial statement preparation procedures; changes in the effective tax rate, which can result from, among other things, changes in the geographic mix of earnings, recent changes in the various and applicable local, federal and international tax laws, or management’s evaluation of the expected outcome of tax disputes. Business and other factors that may cause actual results to vary from expected or historical results include: sales demand; the timing, regulatory approval and commercial success of customers’ products incorporating our products and services, including specifically, the Exubera® Inhalation-Powder insulin device; customers’ changes to inventory requirements and manufacturing plans that alter existing orders or ordering patterns for our products; our ability to pass raw-material cost increases on to customers through price increases; maintaining or improving production efficiencies and overhead absorption; physical limits on manufacturing capacity that may limit our ability to satisfy anticipated demand; the availability of labor to meet increased demand; competition from other providers; average profitability, or mix, of products sold in a reporting period; financial performance of unconsolidated affiliates; strength of the U.S. dollar in relation to other currencies, particularly the Euro, UK Pound, Danish Krone, Japanese Yen and Singapore Dollar; higher interest rates; interruptions or weaknesses in our supply chain, which could cause delivery delays or restrict the availability of raw materials and key bought-in components and finished products, including products produced in northern Israel; raw-material price escalation, particularly petroleum-based raw materials, and energy costs; availability, and pricing of materials that may be affected by vendor concerns with exposure to product-related liability; and, changes in tax law or loss of beneficial tax incentives.
We undertake no obligation to publicly update forward-looking statements, whether as a result of new information, future events or otherwise.

Donald E. Morel, Jr., Ph.D.
Chairman & Chief Executive Officer
William J. Federici
Vice President & Chief Financial Officer
CJS New Ideas Conference
January 11, 2007
NYSE: WST
www.westpharma.com

Certain statements contained in this presentation and certain statements that may be made by management of the
Company orally during this presentation
are forward-looking statements that set forth anticipated results based on
management’s plans and assumptions. Such statements give our current expectations or forecasts of future events – they do
not relate strictly to historical or current
facts. In particular, these include statements concerning future actions, future
performance or results of current and anticipated products, sales efforts, expenses, the outcome of contingencies such as
legal proceedings and financial results. We have
tried, wherever possible, to identify such statements by using words such as
“estimate,” “expect,” “intend,” “believe,” “plan,” “anticipate” and other words and terms of similar meaning in connection
with
any discussion of future operating or financial performance or condition.
We cannot guarantee that any forward-looking statement will be realized. If known or unknown risks or uncertainties
materialize, or if underlying
assumptions are inaccurate, actual results could differ materially from past results and those
expressed or implied in any forward-looking statement. You should bear this in mind as you consider forward-looking
statements. We cannot predict
or identify all such risks and uncertainties, but factors that could cause the actual results to
differ materially from expected and historical results include the following: sales demand; timing and commercial success of
customers’
products incorporating our products and services, including specifically, the Exubera® Inhalation-Powder insulin
device; customers’ changes to inventory requirements and manufacturing plans that alter existing orders or ordering patterns
for
our products; our ability to pass raw-material cost increases on to customers through price increases; maintaining or
improving production efficiencies and overhead absorption; physical limits on manufacturing capacity that may limit our ability
the
availability of labor to meet increased demand; competition from other providers; average profitability, or mix, of products
sold in a reporting period; financial performance of unconsolidated affiliates; strength of the U.S. dollar in relation to other
currencies, particularly the Euro, UK Pound, Danish Krone, Japanese Yen and Singapore Dollar; higher interest rates;
interruptions or weaknesses in our supply chain, which could cause delivery
delays or restrict the availability of raw materials
and key bought-in components and finished products, including products produced in northern Israel; raw-material price
escalation, particularly petroleum-based raw materials, and energy
costs; and availability, and pricing of materials that may be
affected by vendor concerns with exposure to product-related liability; and, changes in tax law or loss of beneficial t
ax incentives.
We undertake no obligation to publicly update forward-looking statements, whether as a result of new information, future
events or otherwise.
Safe Harbor Statement

Corporate Profile
World's premier manufacturer of
components and systems for
injectable drug delivery
Closure systems and prefilled
syringe components
Components for disposable systems
Devices and device sub-assemblies
Founded in 1923
HQ in Lionville, PA
2006 sales $900M (e)
Market capitalization $1.7 Billion as of
December 29, 2006

Diverse Customer Base
Company Estimated Market Share: 70% in Pharma; 70% in Device; 95% in Biotech
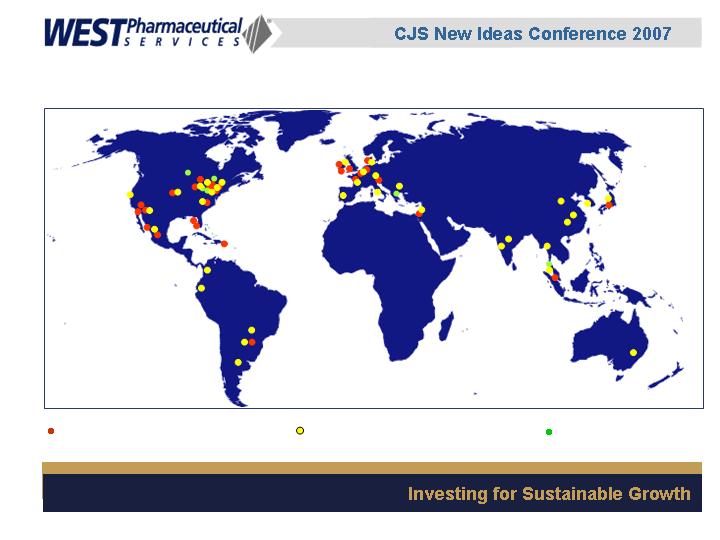
32 manufacturing sites
34 sales offices
7 technical centers
6,000 employees worldwide
Global Presence

Key Company Developments
FY 2001 - 2005
New management team
Divestiture of non-core businesses
Focus returned to injectable packaging and delivery
Increased capital expenditures and capacity build in Europe
Strategic acquisitions
Strong revenue and profit growth
FY 2006
Debt restructuring
Integration of acquisitions
China initiative launched
Second European expansion
Strongest operating year in company history
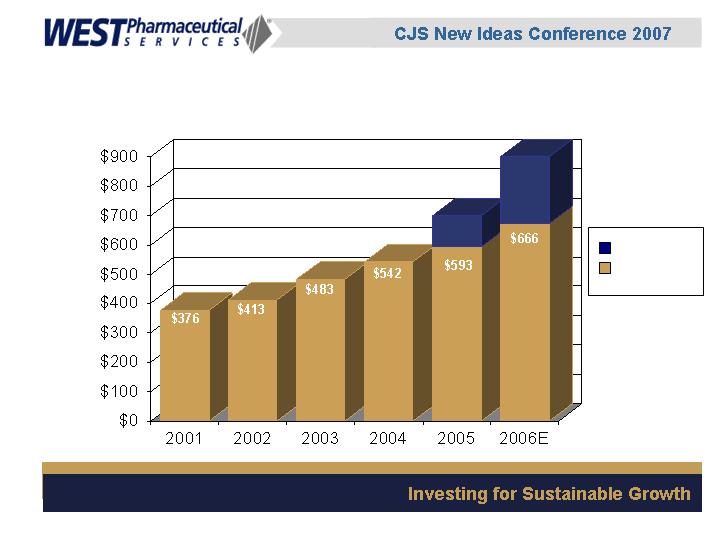
(in millions)
Strong Sales Growth
$224
Acquisitions
Core Business
$107
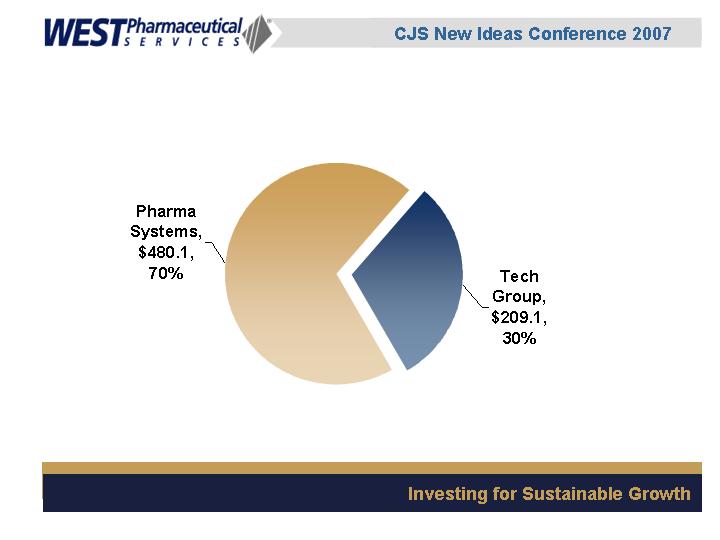
YTD, September 2006
Segment Revenues
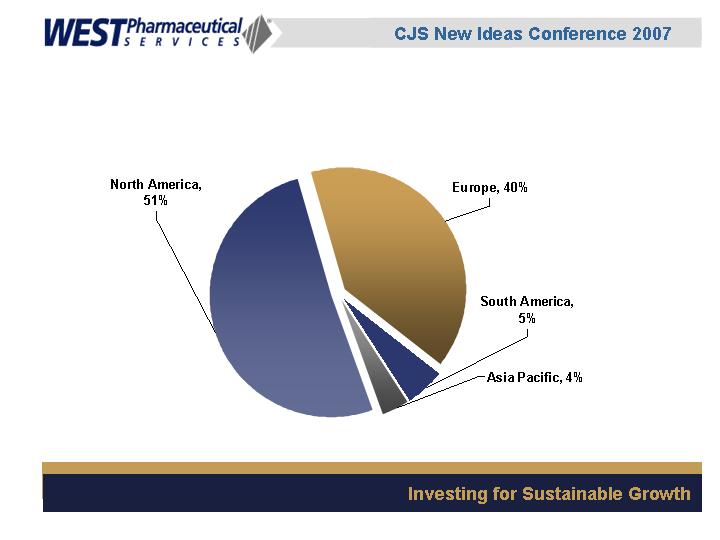
Geographic Sales Mix
(YTD, September 2006)

Corporate Growth Strategy
Pharmaceutical Systems Segment:
Market segmentation
Generate maximum value from key growth drivers
New product innovation
Lean manufacturing
Geographic expansion
Strategic acquisitions
Tech Group Segment:
Leverage West customer base to build market share in multi-
material/multi-component systems for drug administration
Expand proprietary product portfolio through innovation and
strategic technology acquisitions

Market Dynamics Support West’s Continued Growth
Increasing number of patients with chronic illnesses such as diabetes and
cancer
Increasing demand for biologics (2006 Market:$56BN)
Biologics: fastest growing segment of the pharma market out to 2010
(12.2% CAGR)
Injectables currently account of ~15% of the global drug delivery market
Combination products booming
Point of care shift: Hospital Specialty Clinic Home
Parenteral dosage form migration

Dosage Form Migration
Reconstitution
System
Vial – Stopper – Seal
Prefilled Syringe
Ampoule
Auto-injector

West’s Competitive Advantage
Unmatched experience/expertise: drug material interface
Ability to source components from multiple locations globally
Protected IP: West’s components and systems
Regulatory barrier to entry: NDA and ANDA filing must include reference to all
packaging/components in contact with the drug:
1.
West Drug Master File (DMF) 1546 is confidential
2.
West DMF includes functionality data (multi-year studies)
3.
All primary package changes require new stability/ functionality studies
for new filing
Engineering expertise in high volume manufacturing and assembly

FluroTec® and
Barrier Coatings
Westar®
Processing
Strategic Objectives
Continue to maintain market leadership position in the biotechnology space
Components for
Pre-fillable Syringes

Strategic Objectives
Expand position in the insulin/diabetes care segment
Components
for Pen System Applications
Components for
Traditional System
Applications
Devices

Strategic Objectives
Aggressively expand IP position via Innovation and selective acquisitions
Anti-Counterfeiting
Closures
Reconstitution Systems
CZ Resin Systems

Strategic Objectives
Get the global manufacturing footprint issue right

Strategic Objectives
Continue to maintain market leadership position the
biotechnology space
Expand position in the insulin/diabetes care segment
Aggressively expand IP position via Innovation initiative and selective
acquisitions
Get the “manufacturing footprint” issue right
Create financial flexibility through conservative balance
sheet management

Year-to-date September Results
($ Millions)
$1.00
$1.39
E.P.S. Continuing Operations -
Reported
$32.4
$46.8
Income from Continuing Operations
$1.00
$1.50
E.P.S. Continuing Operations - Non
GAAP
$53.2
$80.0
Operating Profit
$87.9
$112.0
SG&A
28%
29%
Gross Margin
$504.0
$681.4
Net Sales
2005
2006
2006 E.P.S. continuing operations – Non GAAP excludes a $0.12 charge related to the refinancing of debt obligations
and a $0.02 favorable tax benefit related to the settlement of a prior year tax claim.

Capital Management
1.79
2.20
Debt to EBITDA Ratio
$46.8
YTD Spending
$90.0
Full Year Estimate
Capital Expenditures:
37.1%
45.0%
Debt to Total Capital
$408.1
$344.0
Total Equity & Minority Interests
$241.1
$281.0
Total Debt
9/30/06
12/31/05
($M)

Focus on core business growth
Segmentation/therapeutic category management
Continue market leadership expansion
Manage through difficult pricing environment
Optimize global capacity utilization
Execute on European capacity expansion and China initiative
Commercialize innovation programs
Operating Priorities

Summary
Start 2007 following a record year
West has a set of competitive advantages that uniquely position
the company for future growth
These competitive advantages capitalize on the primary growth
drivers in key market segments
Global, diverse customer base
Global manufacturing capability
Solid balance sheet
Management incentives closely tied to growth in shareholder
value
Strong corporate governance (ranked #8 in Russell 3000)

Year End Conference Call
Tuesday February 20, 2007 9:00 AM
Review and discussion of 2006 operating results
Business outlook for 2007
Full year earnings guidance
Call in details on website www.westpharma.com

Donald E. Morel, Jr., Ph.D.
Chairman & Chief Executive Officer
William J. Federici
Vice President & Chief Financial Officer
CJS New Ideas Conference
January 11, 2007
NYSE: WST
www.westpharma.com
