UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
FORM 8-K
________________
CURRENT REPORT
PURSUANT TO SECTION 13 OR 15(d)
OF THE SECURITIES EXCHANGE ACT OF 1934
Date of Report (Date of Earliest Event Reported) – May 5, 2006
________________________
WEST PHARMACEUTICAL SERVICES, INC.
(Exact name of registrant as specified in its charter)
_____________________
|
|
|
|
|
Pennsylvania |
1-8036 |
23-1210010 |
|
(State or other jurisdiction |
(Commission File Number) |
(IRS Employer Identification No.) |
|
|
|
|
|
101 Gordon Drive, PO Box 645, Lionville, PA |
|
19341-0645 |
|
(Address of principal executive offices) |
|
(Zip Code) |
610-594-3319
(Registrant’s telephone number, including area code)
Not Applicable
(Former name or address, if changed since last report)
____________________
Check the appropriate box below if the Form 8-K filing is intended to simultaneously satisfy the filing obligation of the registrant under any of the following provisions:
|
o |
Written communications pursuant to Rule 425 under the Securities Act (17 CFR 230.425) |
|
o |
Soliciting material pursuant to Rule 14a-12 under the Exchange Act (17 CFR 240.14a-12) |
|
o |
Pre-commencement communications pursuant to Rule 14d-2(b) under the Exchange Act (17 CFR 240.14d-2(b)) |
|
o |
Pre-commencement communications pursuant to Rule 13e-4(c) under the Exchange Act (17 CFR 240.13e-4(c)) |
Item 7.01 Regulation FD Disclosure
On May 5, 2006, West Pharmaceutical Services, Inc. (the “Company”) issued a press release announcing that the Company will conduct a live webcast of its Investor Day event on Wednesday, May 10, 2006 from 1:00 p.m. to approximately 3:00 p.m. EDT. The event will be held at the Grand Hyatt Hotel in New York, New York. The following members of management will be presenting at the event:
Donald Morel, Chairman and Chief Executive Officer
Steven Ellers, President and Chief Operating Officer
William Federici, Vice President and Chief Financial Officer
Robert Hargesheimer, President, Tech Group
Donald McMillan, President Americas Region, Pharmaceutical Systems
John Paproski, Vice President, Innovation
Michael Schaefers, Director of Marketing, Europe
Mike Anderson, Vice President and Treasurer
A copy of the Company’s presentation from the Investor Day will be available through the Investor link at the Company’s website, http://www.westpharma.com and is also attached hereto as Exhibit 99.1. A copy of the press release is attached hereto as Exhibit 99.2. Both Exhibits are incorporated herein by reference.
The information in this report (including Exhibit 99.1 and 99.2) is being furnished pursuant to Item 7.01 Regulation FD and shall not be deemed to be “filed” for purposes of Section 18 of the Securities Exchange Act of 1934 (the “Exchange Act”) or otherwise subject to the liabilities of that section, nor shall it be deemed to be incorporated by reference in any filing under the Securities Act of 1933 or the Exchange Act.
Item 9.01 Financial Statement and Exhibits
|
(d) |
Exhibits |
|
|
|
|
|
|
|
|
|
Exhibit # |
|
Description |
|
|
99.1 |
|
West Pharmaceutical Services, Inc. presentation slides. |
|
|
|
|
|
|
|
99.2 |
|
West Pharmaceutical Services, Inc. Press Release, dated May 5, 2006. |
SIGNATURE
Pursuant to the requirements of the Securities Exchange Act of 1934, the registrant has duly caused this report to be signed on its behalf by the undersigned hereunto duly authorized.
|
WEST PHARMACEUTICAL SERVICES, INC. |
|
|
|
/s/ John R. Gailey III |
|
John R. Gailey III |
|
Vice President, General Counsel and Secretary |
May 10, 2006

Donald E. Morel, Jr., Ph.D.
Chairman & Chief Executive Officer

Agenda
Don Morel Welcome
Steve Ellers Global Operations Overview
Mike Schaefers Pharma Systems Marketing
Don McMillan Selective Product Overview
Bob Hargesheimer Tech Group
John Paproski Innovation Engine
Bill Federici Financial Overview
All Participants Q&A

Certain statements contained in this presentation and certain statements that may be made by management of the Company orally during
this presentation are forward-looking statements that set forth anticipated
results based on management’s plans and assumptions. Such
statements give our current expectations or forecasts of future events – they do not relate strictly to historical or current facts. In
particular, these include statements
concerning future actions, future performance or results of current and anticipated products, sales
efforts, expenses, the outcome of contingencies such as legal proceedings and financial results. We have tried, wherever possible, to
identify such statements
by using words such as “estimate,” “expect,” “intend,” “believe,” “plan,” “anticipate” and other words and
terms of similar meaning in connection with any discussion of future operating or financial
performance or condition.
We cannot guarantee that any forward-looking statement will be realized. If known or unknown risks or uncertainties materialize, or if
underlying assumptions are inaccurate, actual results could differ
materially from past results and those expressed or implied in any
forward-looking statement. You should bear this in mind as you consider forward-looking statements. We cannot predict or identify all
such risks and uncertainties, but factors
that could cause the actual results to differ materially from expected and historical results
include the following: sales demand; timing and commercial success of customers’ products incorporating our products and services,
including
specifically, the Exubera® Inhalation-Powder insulin device; our ability to pass raw-material cost increases on to customers
through price increases; maintaining or improving production efficiencies and overhead absorption; constraints on manufacturing
capacity in the face of anticipated demand; the availability of labor to meet increased demand; competition from other providers; the
successful integration of acquired businesses; average profitability, or mix, of products sold in a reporting period;
financial performance
of unconsolidated affiliates; strength of the U.S. dollar in relation to other currencies, particularly the Euro, UK Pound, Danish Krone,
Japanese Yen and Singapore Dollar; interruptions or weaknesses in our supply chain, which
could cause delivery delays or restrict the
availability of raw materials and key bought-in components; raw-material price escalation, particularly petroleum-based raw materials,
and energy costs; and availability and pricing of materials that may be
affected by vendor concerns with exposure to product-related
liability.
The Company assumes no obligation to update forward-looking statements as circumstances change. Investors are advised, however, to
consult any further disclosures the Company makes on related subjects
in the Company's 10-K, 10-Q and 8-K reports.
Safe Harbor Statement

Who are we?
Each and every day
millions of West
products are used to
enhance healthcare
around the world.

Corporate Profile
World's premier manufacturer of
components and systems for
injectable drug delivery
Stoppers and seals for vials
Disposable components
used in syringe, IV, blood
collection and diagnostic
systems
Founded in 1923
HQ in Lionville, PA
2005 sales $700M
Market capitalization $1.2 Billion

Diverse Customer Base
Company Estimated Market Share: 70% in Pharma; 70% in Device; 95% in Biotech
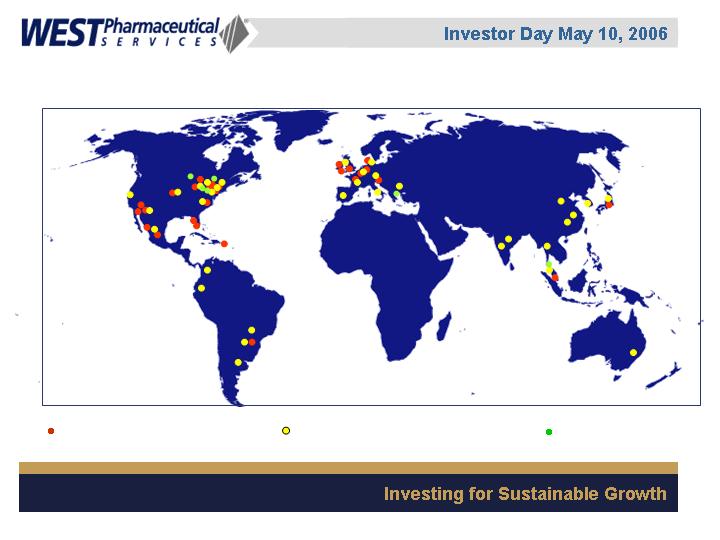
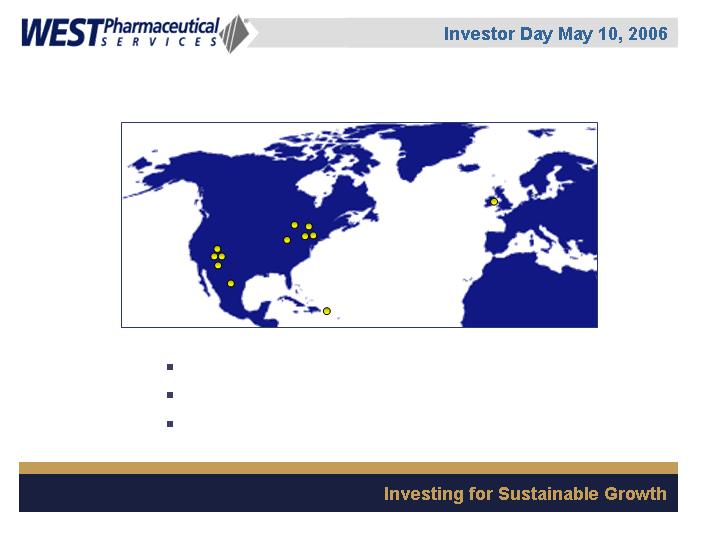
32 manufacturing sites
34 sales offices
7 technical centers
6,000 employees worldwide
Global Presence

Key Company Developments
Acquisition of Monarch Laboratories
Acquisition of The Tech Group
Acquisition of Medimop
Sale of GFI Clinical Unit
Sale of Contract Manufacturing Operations
New management team
Restructuring and increased CAPEX
Sale of OTC Research Services Group
Kinston plant accident
Kinston restart
2-for-1 stock split
Divestiture of Drug Delivery
FY 2005
FY 2004
FY 2003
FY 2002
FY 2001
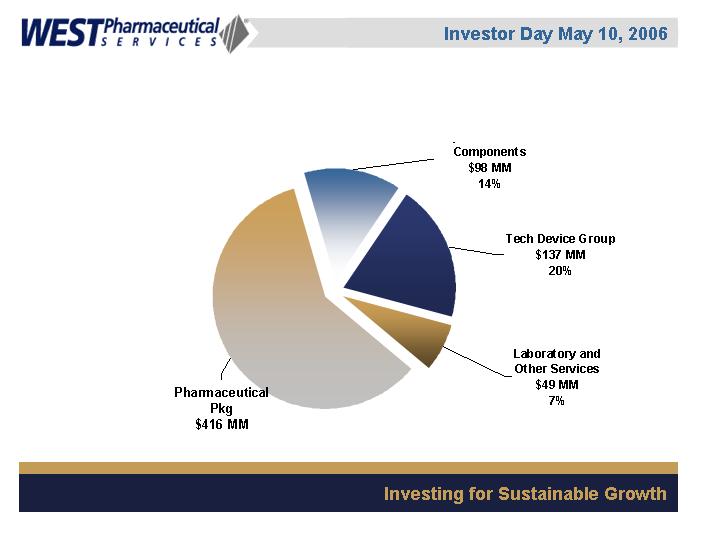
FY05 Net sales: Approx. $700 MM
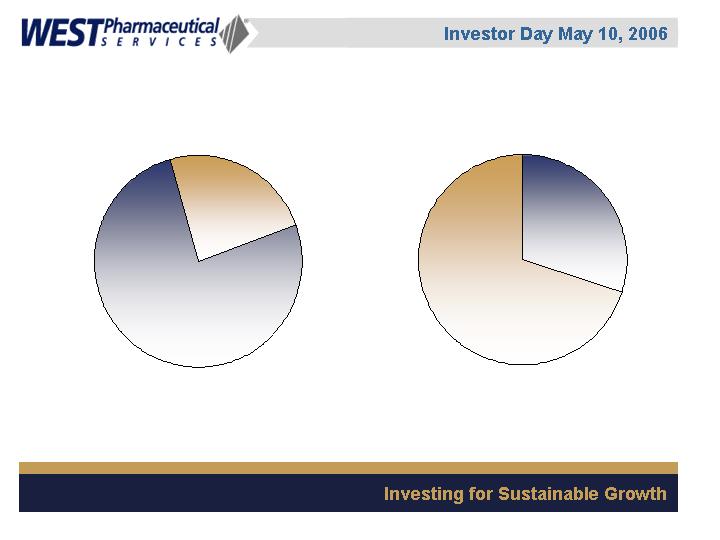
Business Segments
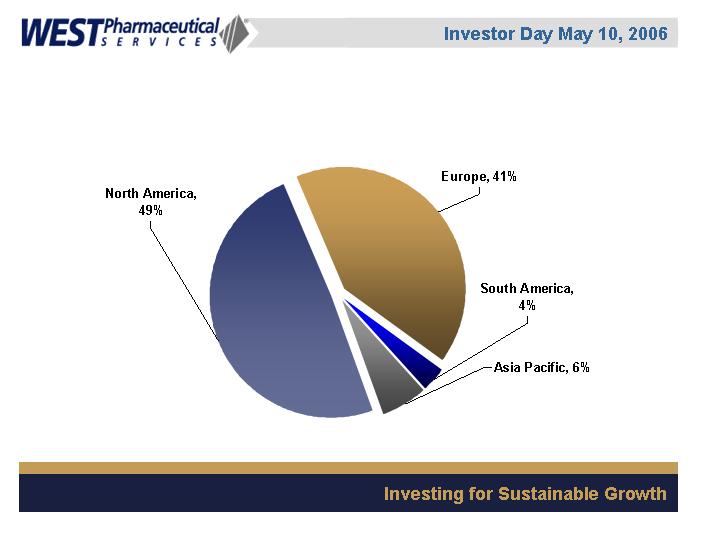
Geographic Sales Mix
(2005, based on point of
sale)

Corporate Growth Strategy
Core Pharmaceutical Systems Business
Maximize the value of West’s core business
(Company estimated market: $1.1 BN)
Market segmentation
Generate maximum value from key growth drivers
New product innovation
Lean manufacturing
Geographic expansion
Strategic acquisitions
Medimop Medical Projects Ltd.

Corporate Growth Strategy
Tech Group Business
Build market share in multi-component systems
for drug delivery
(Company estimated market: $4.5 BN)
Leverage customer base
Develop a portfolio of proprietary systems for injectable,
transmucosal, and pulmonary delivery
Pursue selected consumer opportunities
License or acquire innovative technologies
Pursue strategic acquisitions
The Tech Group

Diabetes
Components
for Pen System Applications
Components for
Traditional System
Applications
Devices

West’s Competitive Edge
Unmatched experience/expertise: drug material
interface
Ability to source
components from multiple
locations globally
Protected IP: West’s components and systems
Regulatory barrier to entry: NDA and ANDA
filing must
include reference to all
packaging/components in contact with the drug:
1.
West Drug Master File (DMF) 1546 is confidential
2.
West DMF includes functionality data (multi-year studies)
3.
All primary package changes require new stability/ functionality
studies for new filing

Investment Considerations
Established platform for sustainable growth
Significant barriers to entry
Favorable growth drivers in key market
segments
Global, diverse customer base
Global manufacturing capability
Strong, experienced management team
Strong corporate governance

Steve Ellers
President & Chief Operating Officer

Overview
Organization
Financial performance
Strategic initiatives
Manufacturing strategy
Product launch process
President and Chief Operating Officer
Business Units with P/L
North America
South America
Europe and Asia Pacific
The Tech Group
Reconstitution (Medimop)
Global Headquarters
Global Supply Chain
& Support Services
Strategic Business
Development
Innovation

2005 Performance
Record sales and profits
Completed three acquisitions
Better positioned for profitable growth
Stronger position with customers
Improved product pipeline
Innovation launched

Drivers for Improved Financial Performance
Returned focus to core injectable business
Reorganized to regional business units
Faster response time to market
Better coordination of resources
Significant increase in CAPEX
Better quality
Greater capacity
Improved efficiencies
Faster response time to market
Value creation
Success with Lean Sigma
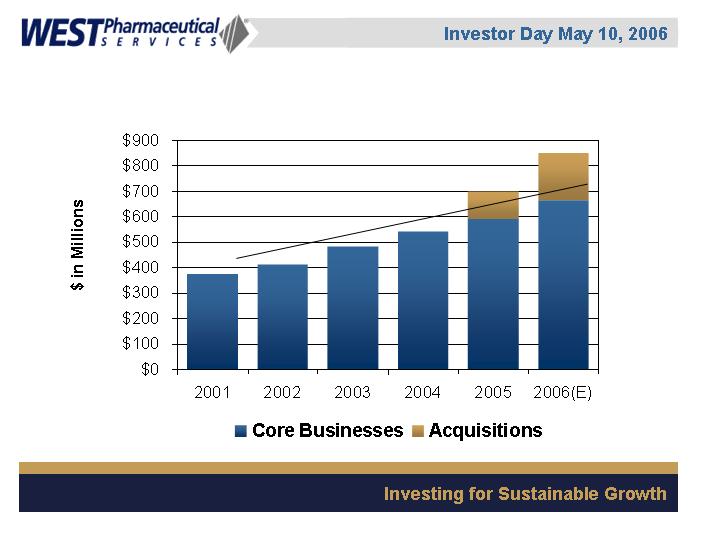
Sales Performance
CAGR = 12%
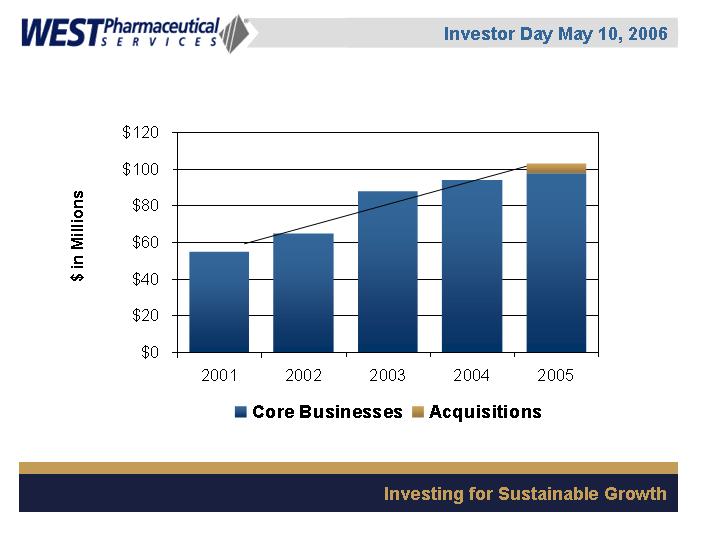
Operating Profit Performance
Note: 2004 operating profit excludes $11.6M of business interruption and out-of-pocket
costs related to the 2003 Kinston accident.
CAGR = 15%
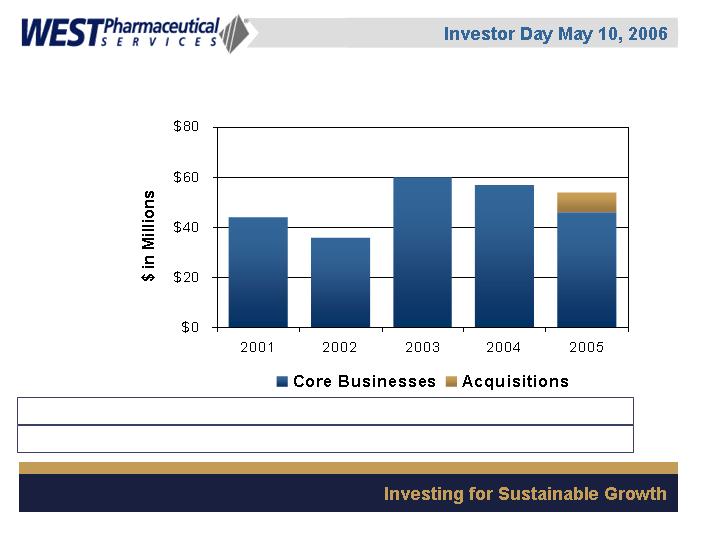
Consolidated Capital Expenditures
Capital as % of sales 12% 9% 12% 11% 8%
Depreciation as % of sales 8% 8% 6% 6% 7%

Key Initiatives For Growth
Market segmentation
Product launches
Accelerate innovation
Lean Sigma initiatives
Expand capacity to meet growing demand

Market Segmentation Identifies Market Needs
Manufacturing Strategy
Capital Expenditures
Product Launches

Market Segment Growth Drivers
Biotechnology/Oncology
Diabetes
Prefillable Syringes
Reconstitution Systems
FluroTec® and Barrier Coatings
Westar®
Processing
West SpectraTM

Market Segment Growth Drivers
Biotechnology/Oncology
Diabetes
Prefillable Syringes
Reconstitution Systems
Components for
Traditional System
Applications
Components
for Pen System Applications

Market Segment Growth Drivers
Biotechnology/Oncology
Diabetes
Prefillable Syringes
Reconstitution Systems

Market Segment Growth Drivers
Biotechnology/Oncology
Diabetes
Prefillable Syringes
Reconstitution Systems

Market Segmentation Identifies Market Needs
Manufacturing Strategy
Capital Expenditures
Product Launches

Manufacturing Philosophy
Centers of Excellence – Focused Factories
Pharmaceutical
Device
Cellular Manufacturing
Zero Defect
Vision inspection
Six Sigma
Multiple site sourcing
Managed as cost centers

Manufacturing Processes
Rubber
Compounding Molding Trimming Finishing
Specialty products
Metal
TrimTec®

Compounding

Molding

Trimming

Washing

Metals

TrimTec®

Market Segmentation Identifies Market Needs
Manufacturing Strategy
Capital Expenditures
Product Launches
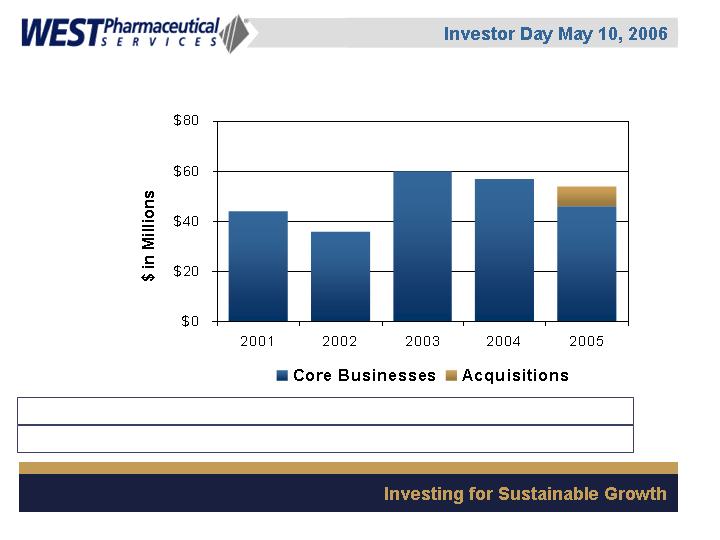
Consolidated Capital Expenditures
Capital as % of sales 12% 9% 12% 11% 8%
Depreciation as % of sales 8% 8% 6% 6% 7%

Market Segmentation Identifies Market Needs
Manufacturing Strategy
Capital Expenditures
Product Launches

Product Launch Process
Match need in market with West product
Target specific customers
Secure development partnerships
Sampling
Machine trials
Stability tests
Regulatory assistance
Analytical Labs

Product Launch Process
Plan – Initiate Launch Campaign
CAPEX investments
Promotion
Training
Sample kits
Broad market - product launch (2 - 3 years later)
Market experience
Market acceptance
Customer ramp-ups

Product Launches
Westar® RS
Elastomer components
Seals
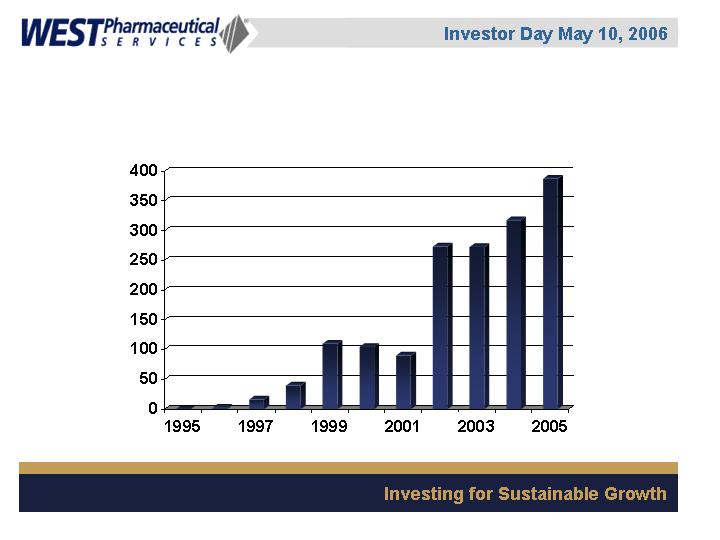
Westar®
Units Sold
(in millions)

Product Launches
Drive development through superior positioning
Investor Day May 10, 2006
FluroTec®
Unrivaled
barrier
coating
Westar®
State of the art
ready-to-sterilize
elastomer
closures
Flip-Off® CCS™
Prewashed and
sterilized
Flip-Off seals
West Spectra™
Covert and overt
security for product
authentication

Key Trends
Growth Drivers
Global demographics – Diabetes
Biotechnology – Oncology, prefilled syringe systems and reconstitution
Anti-counterfeiting needs
Growing demand for capital
Lead market
Add value to customers
Grow faster than market
Cross sell West capabilities
Pharma Systems
Tech
Medimop
Innovation
Innovation creating new markets

Summary
Grow faster than market
Fund CAPEX investments
Accelerate innovation activities:
In house R&D
Licensing
Strategic partnering
Acquisitions
Expand geographically
Synergies from cross selling

Mike Schaefers, Ph.D.
Director of Marketing Europe

Marketing Initiatives
Sustainable above-market growth through:
Global key account management
Marketing initiatives for key growth products
Market segmentation strategy
New product launches / pre-launch activities
Innovations

Global Key Account Management
Based upon well trained and experienced Key
Account Managers
Integrates West’s well-known Technical Support
function into customer relationship
Coordinates all activities between customers and
West globally using the Diamond approach
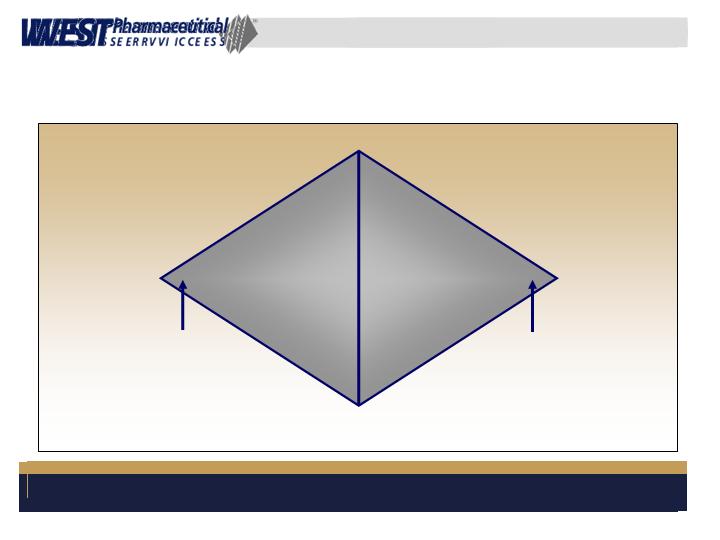
Global Key Account Management – cont.
Marketing
TCS
Regulatory
Operations
QA
R&D
Information
Systems
Investing for Sustainable Growth
Investor Day May 10, 2006
Key Account
Coordination
Supplier
Development
Supplier
Customer
Marketing
Regulatory
Operations
QA
R&D
Information
Systems

Global Key Account Management
Ensures usage of Value Selling / Value Capture
Generates long-term supply agreements
Establishes strategic partnerships with customers

Marketing Initiatives for Growth Products
Product positioning and branding
Focused communication
Sales campaigns
Liaison with opinion leaders, authorities

Marketing Initiatives for Growth Products
Drive development through superior positioning
Investing for Sustainable Growth
Investor Day May 10, 2006
FluroTec®
Unrivaled
barrier
coating
Westar®
State of the art
ready to sterilize
elastomer
closures
Flip-Off® CCS™
Prewashed and
sterilized
Flip-Off seals
West Spectra™
Cover and overt
security for product
authentication

Communication
Develop new, global state-of-the-art web site with links to
key products
Generate and publish product specific articles around key
growth products
Issue ads for key growth products
Exhibit and lecture at key conferences and congresses
globally
Marketing Initiatives For Growth Products

Sales Campaigns
Develop and execute sales campaigns to increase product
awareness and knowledge of customers through:
Mailing to targeted customers
Product specific brochures
Product specific sample kits
Product specific customer presentations
Educational Series
Technical seminars & roadshows
Marketing Initiatives For Growth Products

Strengthen product positioning through liaison with:
Opinion leaders
Professional organizations (i.e. PDA, AAPS )
Regulatory bodies (i.e. FDA, EMEA)
Marketing Initiatives For Growth Products

Implement
Develop
Focus
Market Segmentation Strategy
Analyze market trends and customer needs
Focus development on key segments
Develop segment plans through dedicated, cross-functional
teams
Implement segment specific market plans
Analyze
Investor Day May 10, 2006
Investing for Sustainable Growth

Market Segmentation Strategy
Key growth segments – delivery systems
Prefilled syringes
Medical devices
Key growth segments – markets
Vaccines
Diabetes
Biopharmaceuticals
Generics

Market Segmentation Strategy
Concentrate company and marketing resources
based upon specific market plan i.e.:
Technical Support
Material Sciences
Business Development Manager
Regulatory Affairs
Segment specific sales and communication tools i.e.:
Biotech Sample Kit
Biotech Web Site

Product Launches
Promote reconstitution
and transfer systems
(Medimop)
Clip´n´Ject
Vial Adapter
Mix2Vial
MixJect
Investing for Sustainable Growth
Investor Day May 10, 2006

Product Launches
Prefilled syringe
component offerings
Complete range of
FluroTec plungers from
0.5 ml to 10 ml
Global launch of new,
´state of the art´
Thermoset Formula for
Needle Shields and Tip
Caps
Launch FluroTec Tip Cap
into market
Investing for Sustainable Growth
Investor Day May 10, 2006

Six Sigma Quality Product
Meeting customer demand for enhanced quality products
100 % inspected
Low particulate matter
Low protein contamination
Product Launches

Summary
Sustainable above-market growth through:
Global key account management
Marketing initiatives for key growth products
Market segmentation strategy
New product launches / pre-launch activities
Innovations

Don McMillan
President, North America
Pharmaceutical Systems Division

Pharmaceutical and Biotech Market
Pharmaceutical Market
West has strong position for packaging components
Low single digit growth rates
Focusing on Biotech to strengthen their pipelines
Partnership, acquisitions and alliances
Biotech Market
CAGR 15% (2003 – 2008)
High value products
Sensitive proteins and peptides
Targets for counterfeit drugs
Predominately injectable products
West products are low percentage of COGS
West enjoys “partnership” relationships
Many emerging companies
West’s focus on the Biotech Market
Value drivers
Drug product efficacy
Management of risk
Patient convenience and compliance
Product R&D driven by the needs of the Biotech market
Proprietary products
Value added products drive growth in excess of volume
Incorporate advanced technologies
Barrier coatings for elastomers
Westar processing of elastomers
Clinical to commercial standardization
Compliant with latest regulatory requirements and standards
Customized products for anti-counterfeit protection

West FluroTec

Westar

Product Evolution to Combat Drug Counterfeiting
Permission granted by Amgen, Inc.
West’s standard product
2nd generation product
3rd generation product
2nd generation product

The Next Generation Products
Package authentication with overt and covert technologies
Item level serialization for electronic track and trace

Emerging Biotech
Many small companies focused on bringing new therapies to
market
West has an opportunity to:
Educate the market
Share regulatory and packaging science expertise
Dedicated Emerging Biotech team
Consultative selling process
Tailor products to their specific needs
Laboratory services
Packaging components
Introduce next generation products and services
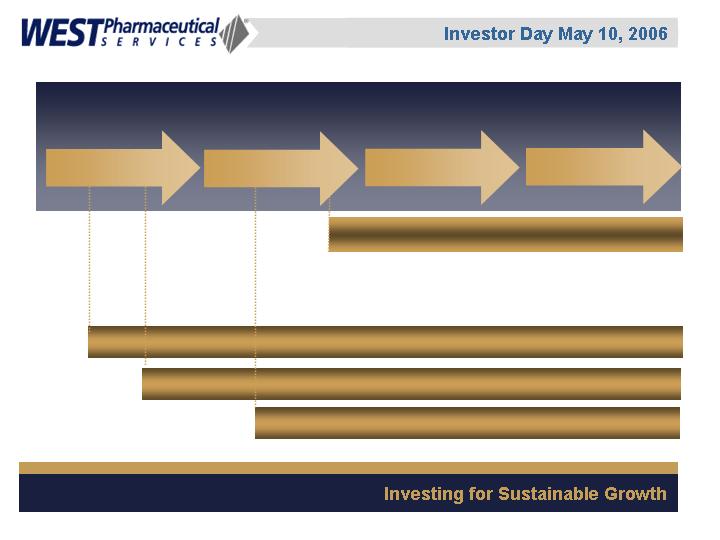
Customers’ Product Development Cycle
Laboratory Services
Delivery/Device Systems
Packaging Components
Traditional West Interface:
Packaging Components
West’s Evolving Interface:
Phase III
Commercialization
Phase II
Phase 1

Conclusion
Existing relationships with major players
Dedicated effort for emerging companies
Broad line of existing proprietary products and
services
Biotech market needs drive product R&D
Leverage market knowledge to identify and introduce
additional value added products and services

Bob Hargesheimer
President, The Tech Group

Custom injection molding and assembly services
focused on OEM customers in the
consumer and health care markets.
Investing for Sustainable Growth
Acquired by West, May 2005
The Tech Group
FY04 Sales: $74.2 MM
Consumer
24%
Health Care 76%
West Device Group
FY04 Sales: $65.0 MM
Health Care
30%
Consumer 70%

The Tech Group Offerings
Custom contract-manufacturing services
Precision plastic injection-molding and assembly
Expertise in product design, including in-house mold
design and construction
Automated assembly solutions
Quick-response center for developmental and
prototype tooling

Markets
Health Care
Consumer

Customer Base
Health Care
Device
Consumer
Internationally Positioned
12 manufacturing facilities
1,900 employees
887,000 square feet for manufacturing

The Tech Group Growth Strategy
Market leadership through technical
excellence.
Design services
Regulatory expertise
Analytical lab services
Tooling
High volume assembly
Six Sigma support

The Tech Group Growth Strategy
Build share in multi-component systems.
Leverage relationships and experience.
Add value with West experience/capabilities.
Technology support for proprietary systems.
Pursue select Consumer opportunities.
Technology based advantage.

Health Care Market Segments
Pharmaceutical Delivery
Surgical/Medical Device

Health Care Market Segments
Diagnostic
Other

The Tech Group Growth Strategy
Build share in multi-component systems.
Leverage relationships and experience.
Add value with West
experience/capabilities.
Technology support for proprietary
systems.
Pursue select Consumer opportunities.
Technology based advantage.

Consumer

Summary
Continued growth through:
Building share in multi-component systems
Pursue selected consumer opportunities
Focus on key services
Leverage West and Tech capabilities
Execute on product pipeline

Investing for Sustainable Growth
John Paproski
Vice President of Innovation

Focus On Growth: Historical View
Drug Development Division was a focal point
Formulation and Nasal Based Delivery Systems -- exited in 2005
Plastic Division (now Tech Group) added new processing technologies
State-of-art molding technologies
Clean room molding
High speed assembly
Market the capability as a custom molder
Core Injectable business focused on value-added features to
proprietary products
Daikyo technologies and Japanese Quality
Westar
Coatings
Seals
Prefilled syringe components

Focus On Growth: Historical View: R&D
Global Engineering and Product Development
Product design and development
Tooling design and construction
Elastomer material development
Design and construction of assembly and processing equipment
Develop new packaging and device concepts
# of Employees: 64 professionals in these activities.
Segment Expenditures
2005 2004 2003
Pharma Systems $6.3 MM $5.2 MM $4.6 MM
Tech Group $1.6 MM $1.6 MM $1.7 MM

Innovation at West: Stepping It Up
Refocused on proprietary products for injectable
drug packaging and delivery systems
Increased innovation spending an additional $4-6M in
2006
Multiple technology license/agreements acquired
Increased I.P. activity
Prototype thru scale up developments
Medimop acquisition
Established an innovation process and group
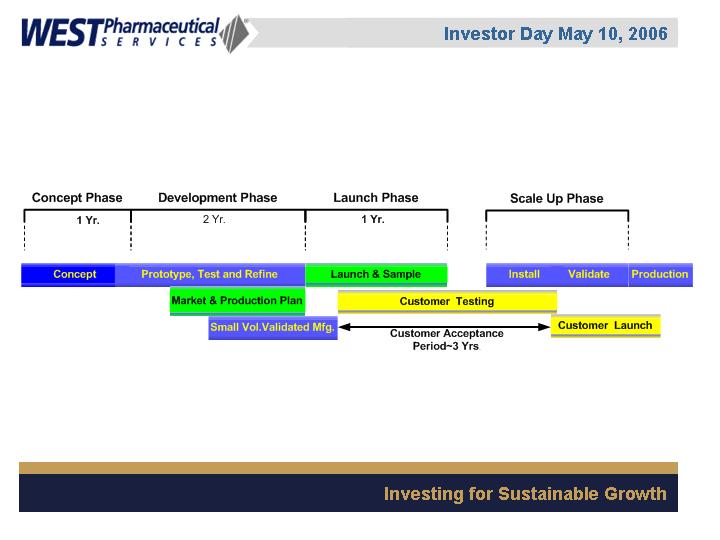
Innovation at West: The Process

Required Elements for Innovation Success
1.
Market need
2.
Customer access
Current Focus: The Pharmaceutical Market
3.
West core competency
Injectable technical expertise and support
Global manufacturing capability in metal, rubber,
plastic and assembly
GMP and GLP capabilities
Creative injectable product developer
4.
Technology match

Injectable Pharma Market Trends
Sustained new biotech NME’s
Frequent lyophilized NME’s
Prefilled syringe growth
Self administration
Patient/clinician convenience as a differentiator
Simplify pharma filling and processing
Needle safety
Counterfeit concerns
Long acting drug formulations
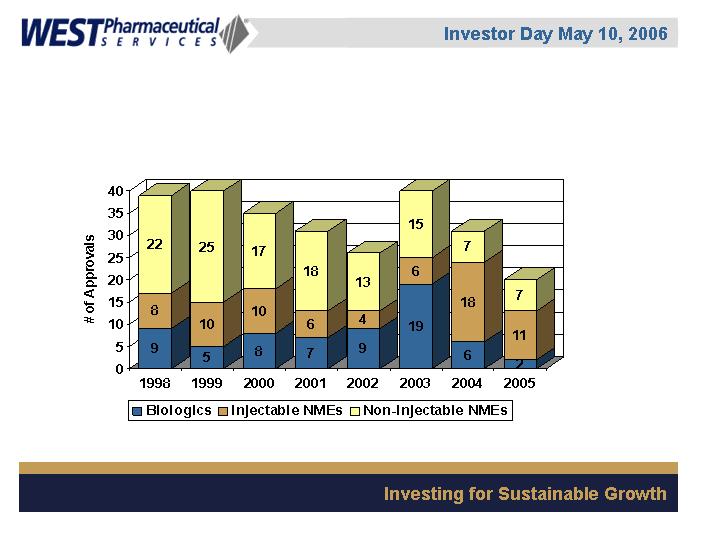
Innovation at West: Importance of Injectables
NME & BLA Approvals (FDA)
Source: The Pink Sheet; FDA Website
39
40
35
31
26
40
31
20

Therapeutic Areas
Anti-Infectives
Biologicals
Cardiovascular
Diabetes Therapy
Hormones
Immunology
Neurology
Oncology
Ophthalmology
Osteoporosis
Market Trends
Pre Filled Syringe Growth
Self Administration
Abundant Biotech NME’s
Frequent Lyophilized NME’s
Needle Safety
Patient/Clinician Convenience as a
Differentiator
Simplify Pharma Filling and Processing
Long Acting Drug Formulations
Counterfeit Concerns
Therapeutic
Categories
Pharma Market
Trends

The Reconstitution Product Category: Market Drivers
Lyophilized NME’s
Necessity for stability
Self administration
Caregiver convenience: a pharma product differentiator

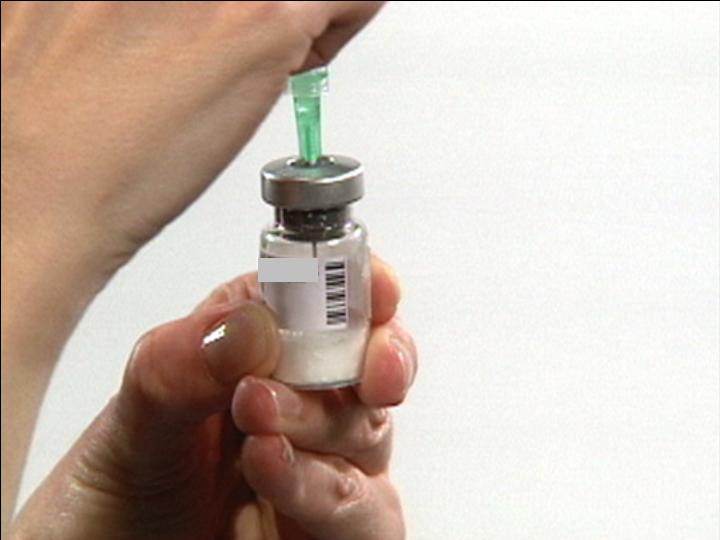
Reconstitution Supplies

Remove Flip Off™
From Diluent Vial

Remove Flip Off™
From Drug Vial

Alcohol Swab
Stopper Surface
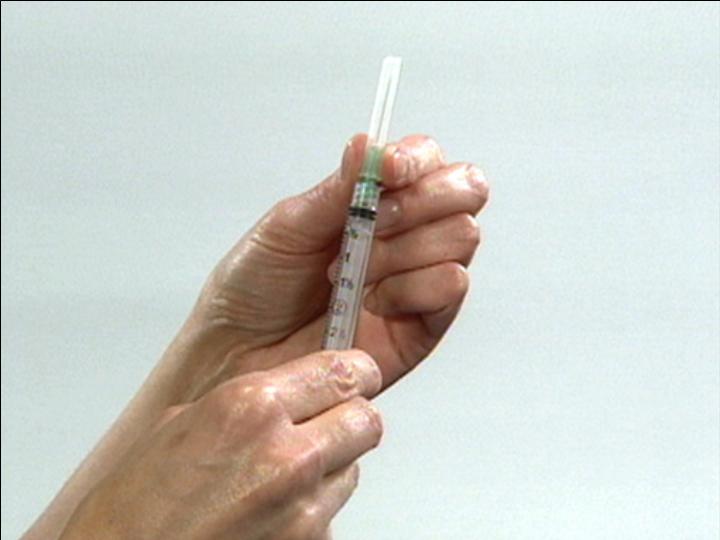
Remove Needle Cover
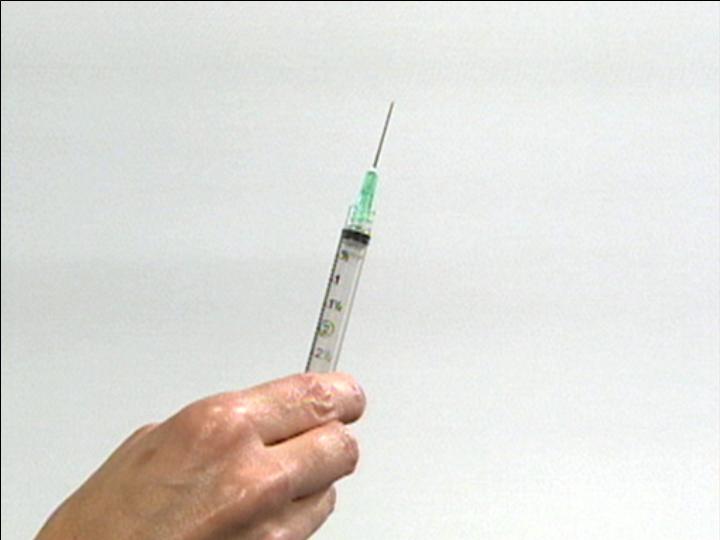
Prepare Syringe
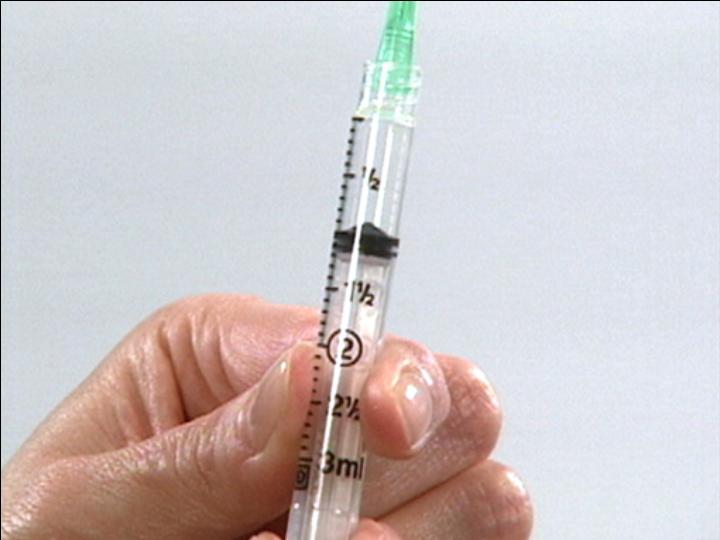
Draw Air Into Syringe
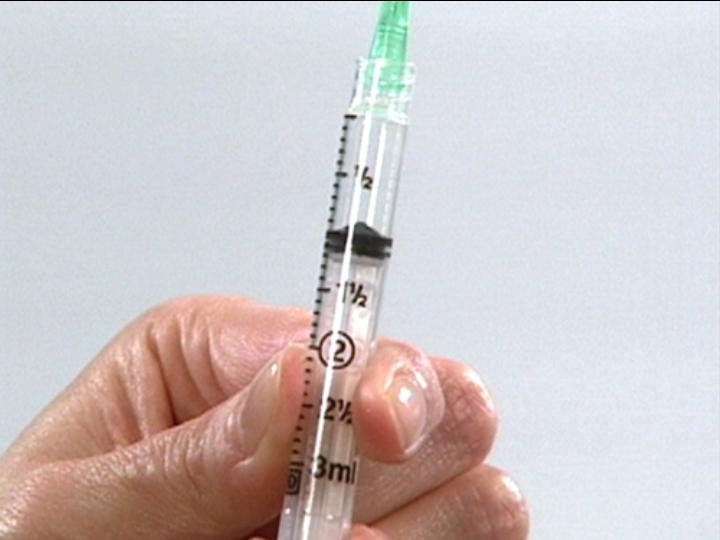
Prepare for
Dilluent Withdrawl
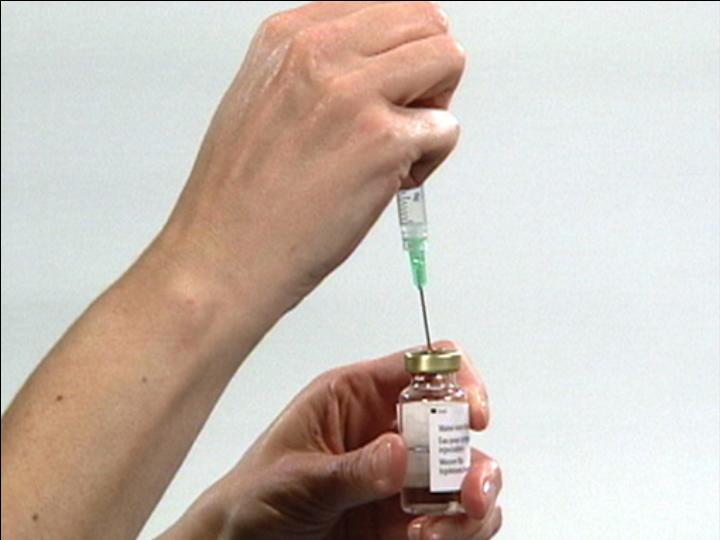
Insert Syringe into
Vial Stopper
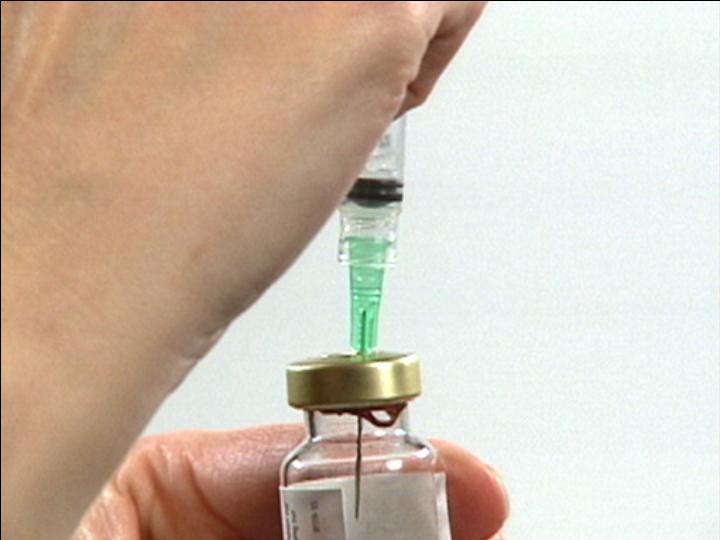
Push Air into Vial
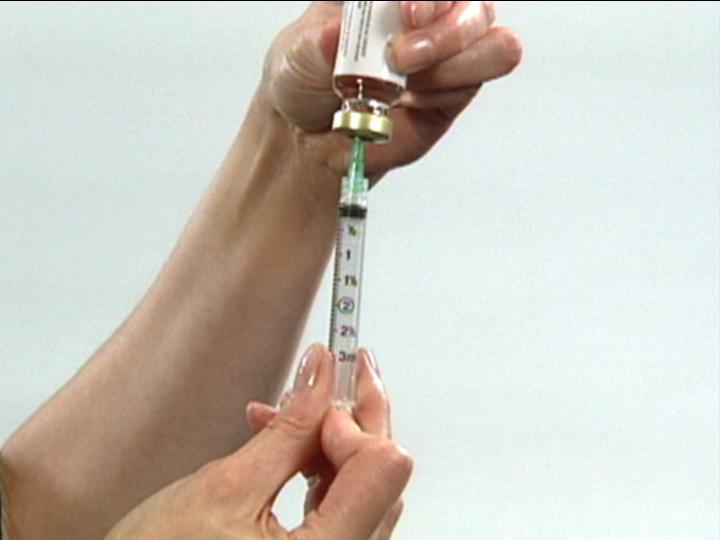
Withdraw Dilluent
From Vial
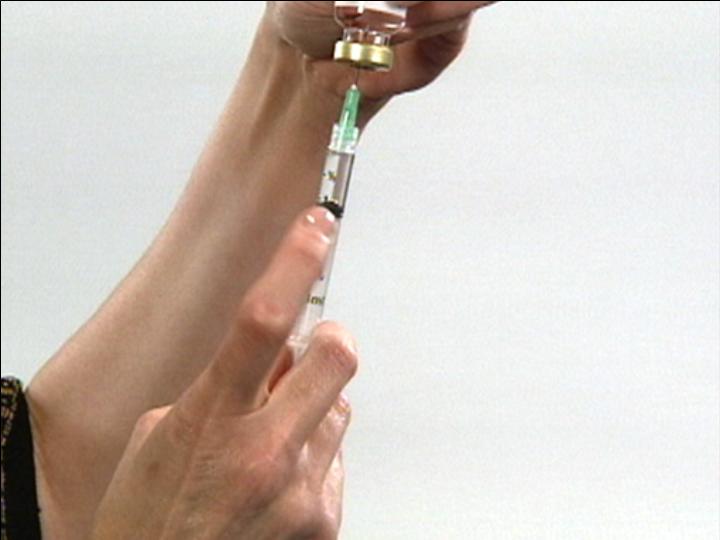
Adjust Dose in Syringe
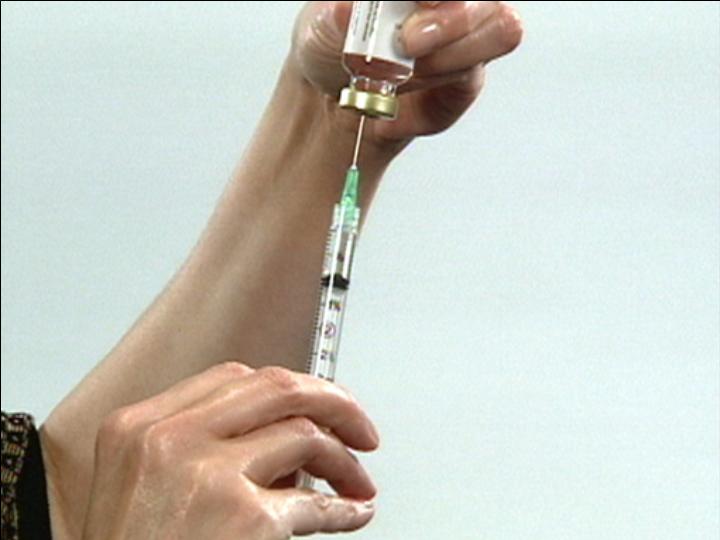
Withdraw Syringe
from Vial
Insert Syringe into
Drug Vial
Inject Liquid Into
Drug Vial
Unmixed Solution

Dispose of Syringe

Mix Solution
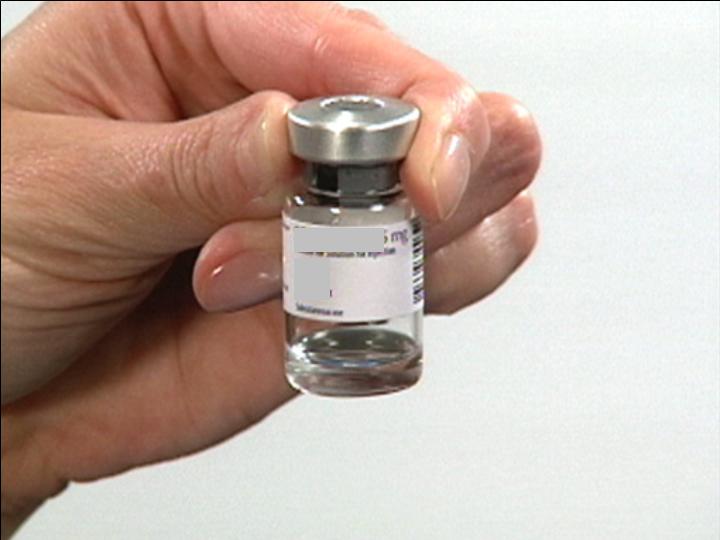
Insure Mixing is Complete
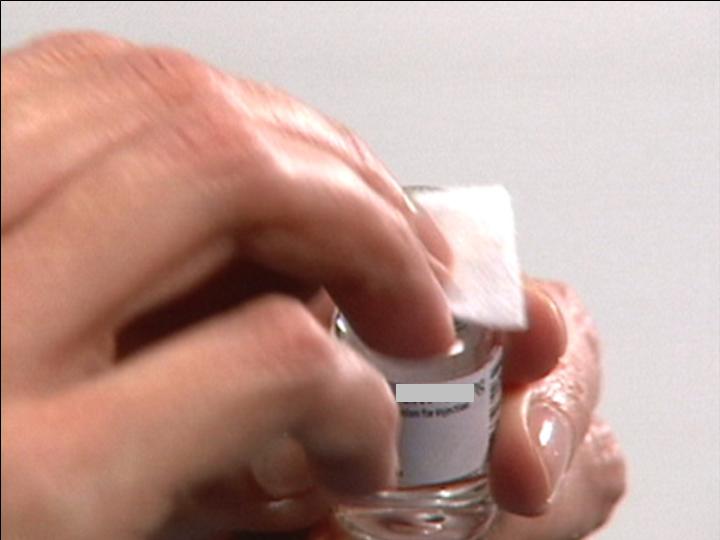
Swab the top of
the Drug Vial
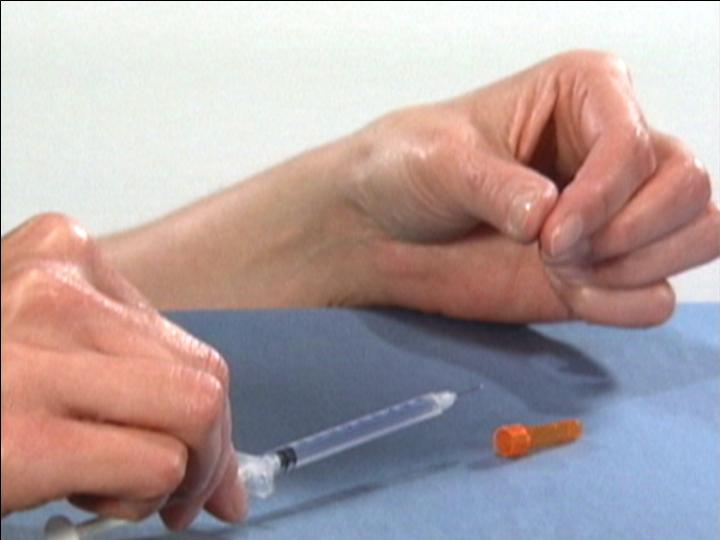
Prepare the Injection Syringe
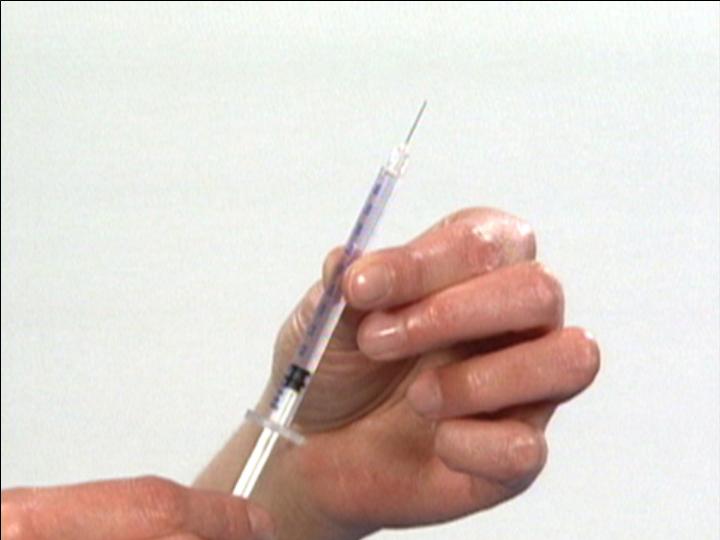
Draw air into the
Injection Syringe
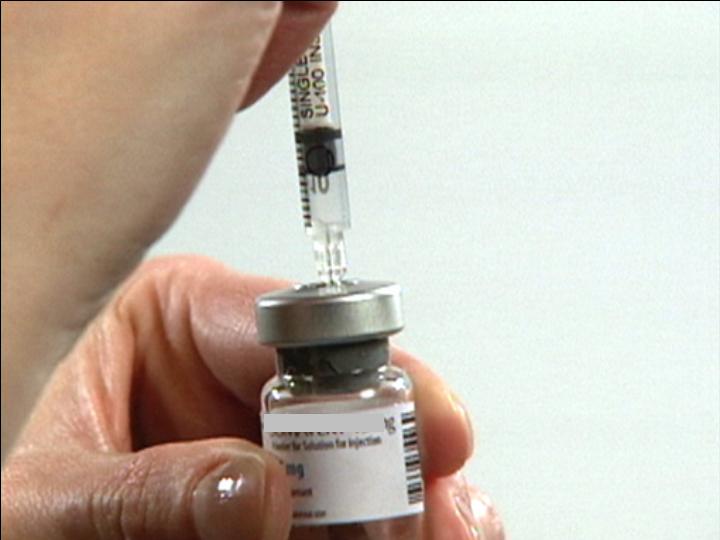
Push air into the Vial
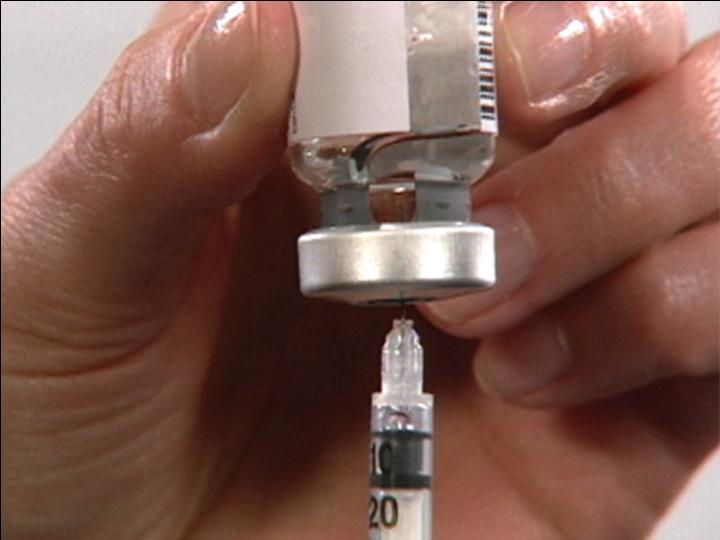
Invert Vial
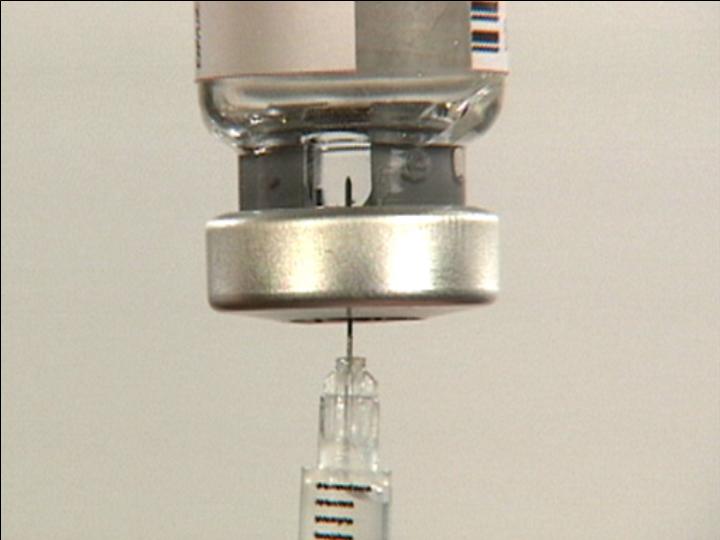
Carefully
Position
Needle
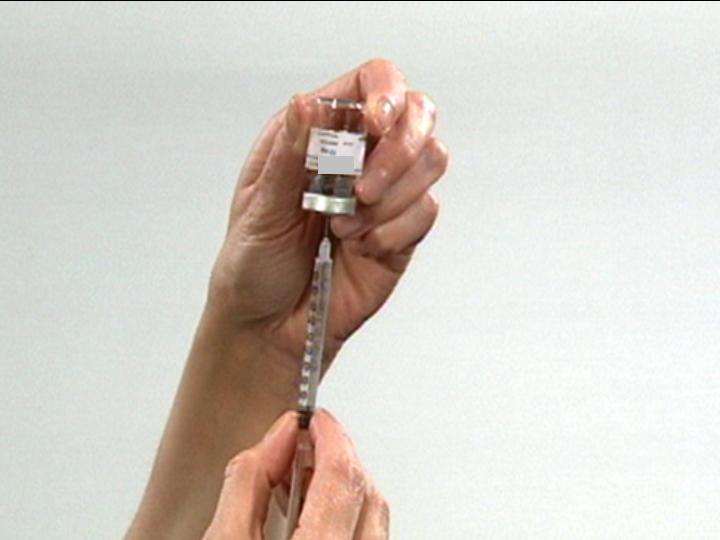
Draw Contents from Vial
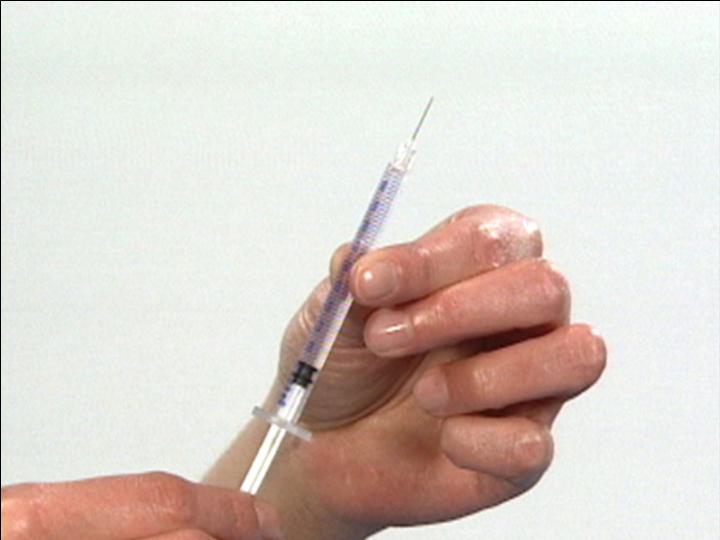
Ready for Injection

The Reconstitution Product Category
Innovation at West: Portfolio Building
West’s Traditional Products
Clip’n’Ject
Medimop
Targeting Innovation
ALIGN
PRODUCT CATEGORY WITH MARKET TRENDS
Vial Projects
Component Projects
To Be Announced
To Be Announced
Reconstitution
Counterfeiting
Pharma
Processing
Caregiver
Convenience
Caregiver
Safety
Lyo NME’s
Biotech NME’s
Self Admin.
PFS Growth
Inj. Pharma
Market
Trends
Product
Category

Innovation at West:
Growing New Product Portfolio
Expansion within the product category
Spin-off products
Evolutionary path within the category
Follow-up product generations
Supporting products
New categories
Focused and manageable injectable projects
Future non injectable
Cross category

Innovation Summary
Best pipeline of products in recent history
Offerings will:
Relate directly to customers and markets we now serve
Leverage the combined capabilities and customer access
that exist in our:
Divisions
Partners
Recently acquired businesses

William Federici
Chief Financial
Officer
Investing for Sustainable Growth

.12
-
-
-
-
-
Interest Payment
$.54*
$1.40
$1.33
$1.32
$.96
$.83
EPS Non-GAAP
-
-
(.02)
-
-
-
Affiliate Gain
-
-
.30
(.42)
-
-
Kinston
-
-
-
-
(.03)
-
Foreign Currency
(.01)
.05
(.07)
-
(.09)
-
Unusual Tax
Items
-
(.04)
.03
.26
.30
.05
Restructuring
$0.43
$1.39
$1.09
$1.48
$.78
$.78
EPS Reported
Q1 2006
2005
2004
2003
2002
2001
GAAP/Non-GAAP Reconciliation
* Included in 2006 full-year non GAAP EPS guidance of $1.68 – $1.78.
This information is being provided for the convenience of readers in making comparisons between the periods presented
and other periods
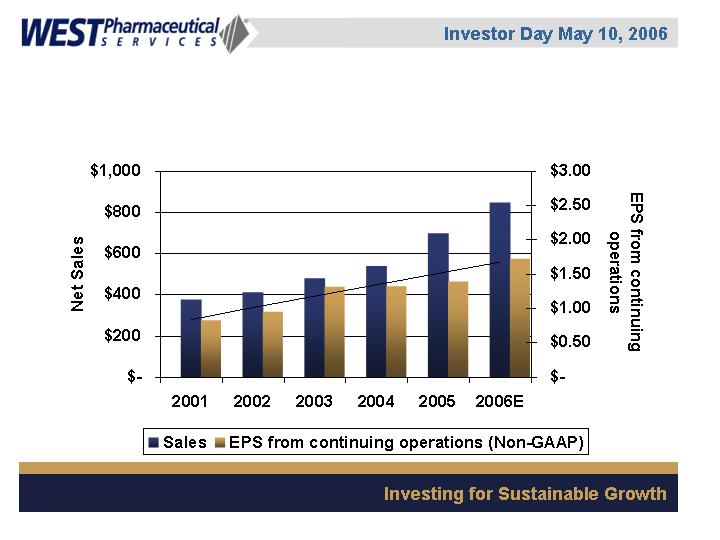
Consolidated Sales and Non-GAAP EPS
from Continuing Operations
($ in millions)

First Quarter Results
($ in millions, except per share data)
$0.41
$0.43
E.P.S. from Continuing Operations
$0.41
$0.54
Non-GAAP E.P.S. from Continuing Operations
$13.0
$14.3
Income from Continuing Operations
$20.1
$28.2
Operating Profit
$25.2
$38.0
Selling, General & Administrative Expenses
31%
30%
Gross Margin
$149.5
$222.8
Net Sales
2005
2006
2006 E.P.S. from continuing operations includes a $0.12 charge related to the refinancing of senior
notes and a $0.01 favorable impact related to an IRS tax claim settlement.

Capital Management
$118.7
$133.5
Working Capital
$11.4
YTD Spending
Capital Expenditures:
45.0%
42.3%
Debt to Total Invested Capital
$625.0
$640.6
Total Capitalization
$281.0
$271.0
Total Debt
12/31/05
3/31/06
($M)
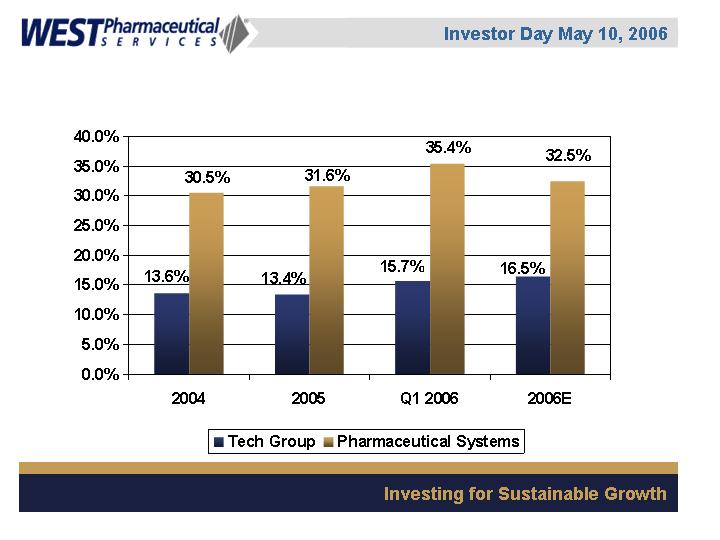
Gross Margin Percentages
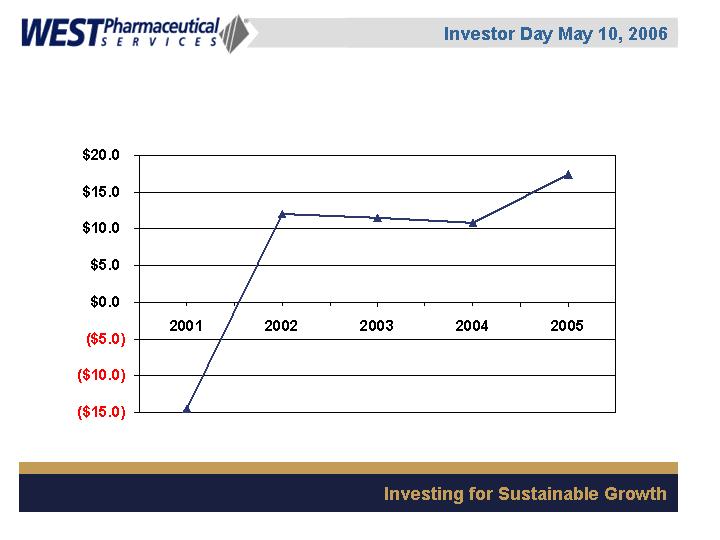
Corporate Cash Flow
($ in millions)
Corporate Cash Flow is defined as operating cash flow less capital expenditures and dividends.
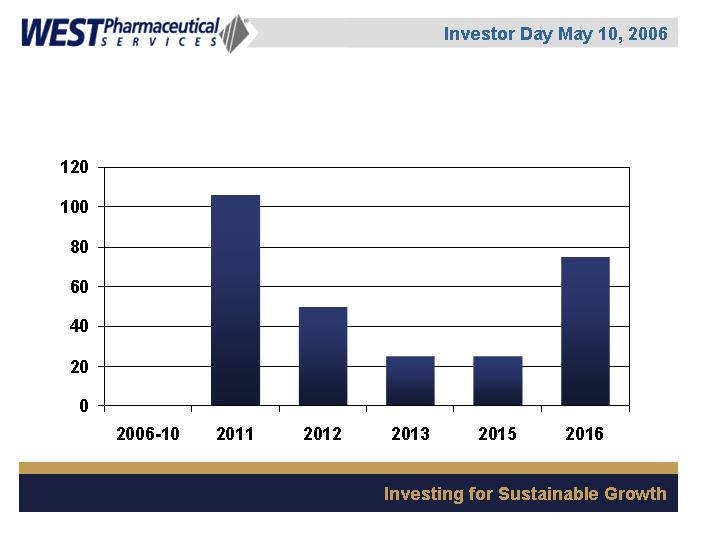
Debt Maturity Schedule
($ in millions)
$0
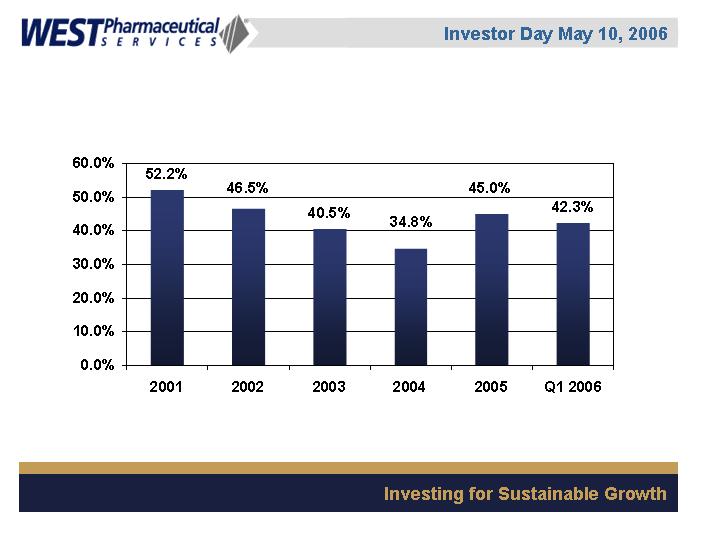
Debt to Total Capitalization
Goal: 40-45% excluding acquisitions and significant capital expansion

In Summary
Strong cash flow from operations
Strong balance sheet
Foundation is in place to fund future growth
Focus on cost control and lean initiatives

Donald E. Morel, Jr., Ph.D.
Chairman & Chief Executive Officer

Summary
West has a set of competitive advantages that
uniquely position the company for future growth
These competitive advantages capitalize on the
primary growth drivers in key market segments
Global, diverse customer base
Solid financial base
Strong, experienced management team focused
on execution

|
Contacts: West Pharmaceutical Services, Inc. Michael A. Anderson Vice President and Treasurer (610) 594-3345 |
Investors/Financial Media: Financial Dynamics Julie Huang/Theresa Kelleher (212) 850-5600 wst@fd-us.com |
Exhibit 99.2
West Pharmaceutical Services to Host Investor Day
- Wednesday, May 10 at the Grand Hyatt in New York City -
LIONVILLE, Pa., May 5 /PRNewswire-FirstCall/ -- West Pharmaceutical Services, Inc. (NYSE: WST), the global market leader in closure systems and syringe components for use with injectable drugs, announced today that it will conduct a live webcast of its Investor Day event on Wednesday, May 10, 2006 from 1:00 p.m. to approximately 3:00 p.m. EDT. The event will be held at the Grand Hyatt Hotel in New York, New York.
The following members of management will provide an in-depth look at the Company’s global operations, business segments, supply chain management and investments in innovation. Speakers will include:
|
|
• |
Donald Morel, Chairman and CEO |
|
|
• |
Steven Ellers, President and COO |
|
|
• |
William Federici, Vice President and CFO |
|
|
• |
Robert Hargesheimer, President, Tech Group |
|
|
• |
Donald McMillan, President Americas Region, Pharmaceutical Systems |
|
|
• |
John Paproski, Vice President, Innovation |
|
|
• |
Michael Schaefers, Director of Marketing, Europe |
|
|
• |
Mike Anderson, Vice President and Treasurer |
A live audio portion and a copy of the presentations will be available through the Investor link of the Company’s website, http://www.westpharma.com. A replay of the audio portion will be available for 14 days. To access the live webcast, visit the company’s Web site at least 15 minutes prior to the scheduled start time to download any necessary audio or plug-in software.
About West Pharmaceutical Services, Inc.
West Pharmaceutical Services, Inc. is the world’s premier manufacturer of components and systems for injectable drug delivery, including stoppers and seals for vials, and closures and disposable components used in syringe, IV and blood collection systems. The Company also provides products with application to the personal care, food and beverage markets. West’s customers include the world’s leading pharmaceutical, biotechnology, generic drug and medical device producers. Headquartered in Lionville, Pennsylvania, West Pharmaceutical Services supports its partners and customers from 50 locations throughout North America, South America, Europe, Mexico, Japan, Asia and Australia. For more information, visit West at http://www.westpharma.com.
SOURCE West Pharmaceutical Services, Inc.
CONTACT: Michael A. Anderson, Vice President and Treasurer of West Pharmaceutical Services, Inc., +1-610-594-3345; or Investors-Financial Media: Julie Huang or Theresa Kelleher, both of Financial Dynamics, +1-212-850-5600, wst@fd-us.com, for West Pharmaceutical Services, Inc.

